Full List of Publications: Google Scholar
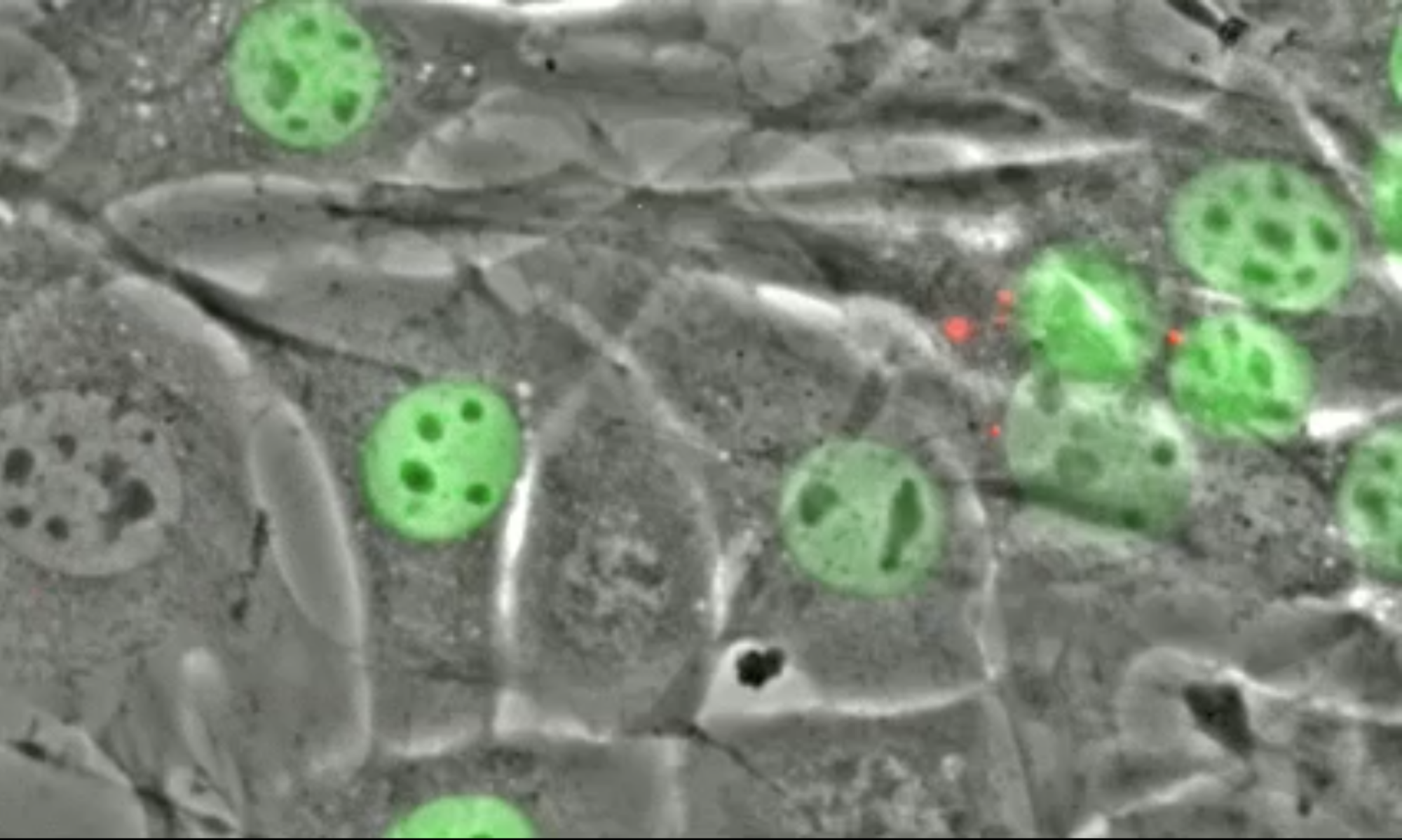
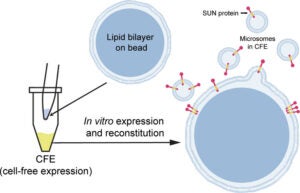 S Majumder, YY Hsu, H Moghimianavval, M Andreas, TW Giessen, GWG Luxton, AP Liu (2022), In Vitro Synthesis and Reconstitution Using Mammalian Cell-Free Lysates Enables the Systematic Study of the Regulation of LINC Complex Assembly. Biochemistry.
S Majumder, YY Hsu, H Moghimianavval, M Andreas, TW Giessen, GWG Luxton, AP Liu (2022), In Vitro Synthesis and Reconstitution Using Mammalian Cell-Free Lysates Enables the Systematic Study of the Regulation of LINC Complex Assembly. Biochemistry.
Understanding the structure and structure–function relationships of membrane proteins is a fundamental problem in biomedical research. Given the difficulties inherent to performing mechanistic biochemical and biophysical studies of membrane proteins in vitro, we previously developed a facile HeLa cell-based cell-free expression (CFE) system that enables the efficient reconstitution of full-length (FL) functional inner nuclear membrane Sad1/UNC-84 (SUN) proteins (i.e., SUN1 and SUN2) in supported lipid bilayers. Here, we provide evidence that suggests that the reconstitution of CFE-synthesized FL membrane proteins in supported lipid bilayers occurs primarily through the fusion of endoplasmic reticulum-derived microsomes present within our CFE reactions with our supported lipid bilayers. In addition, we demonstrate the ease with which our synthetic biology platform can be used to investigate the impact of the chemical environment on the ability of CFE-synthesized FL SUN proteins reconstituted in supported lipid bilayers to interact with the luminal domain of the KASH protein nesprin-2. Moreover, we use our platform to study the molecular requirements for the homo- and heterotypic interactions between SUN1 and SUN2. Finally, we show that our platform can be used to simultaneously reconstitute three different CFE-synthesized FL membrane proteins in a single supported lipid bilayer. Overall, these results establish our HeLa cell-based CFE and supported lipid bilayer reconstitution platform as a powerful tool for performing mechanistic dissections of the oligomerization and function of FL membrane proteins in vitro. While our platform is not a substitute for cell-based studies, it does provide important mechanistic insights into the biology of difficult-to-study membrane proteins.
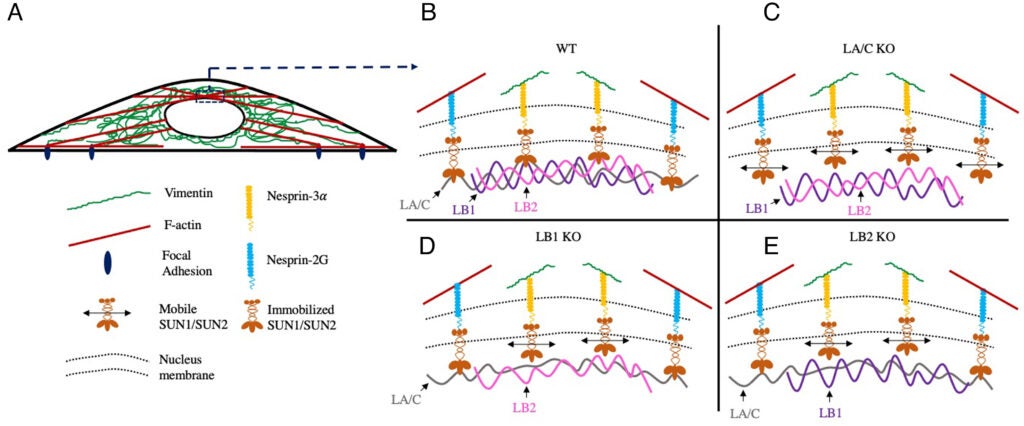 A Vahabikashi, S Sivagurunathan, FA Sadsad Nicdao, YL Han, CY Park, M Kittisopikul, X Wong, JR Tran, GG Gundersen, KL Reddy, GWG Luxton, M Guo, JJ Fredberg, Y Zheng, SA Adam, RD Goldman (2022), Nuclear lamin isoforms differentially contribute to LINC complex-dependent nucleocytoskeletal coupling and whole-cell mechanics. PNAS.
A Vahabikashi, S Sivagurunathan, FA Sadsad Nicdao, YL Han, CY Park, M Kittisopikul, X Wong, JR Tran, GG Gundersen, KL Reddy, GWG Luxton, M Guo, JJ Fredberg, Y Zheng, SA Adam, RD Goldman (2022), Nuclear lamin isoforms differentially contribute to LINC complex-dependent nucleocytoskeletal coupling and whole-cell mechanics. PNAS.
The ability of a cell to regulate its mechanical properties is central to its function. Emerging evidence suggests that interactions between the cell nucleus and cytoskeleton influence cell mechanics through poorly understood mechanisms. Here we conduct quantitative confocal imaging to show that the loss of A-type lamins tends to increase nuclear and cellular volume while the loss of B-type lamins behaves in the opposite manner. We use fluorescence recovery after photobleaching, atomic force microscopy, optical tweezer microrheology, and traction force microscopy to demonstrate that A-type lamins engage with both F-actin and vimentin intermediate filaments (VIFs) through the linker of nucleoskeleton and cytoskeleton (LINC) complexes to modulate cortical and cytoplasmic stiffness as well as cellular contractility in mouse embryonic fibroblasts (MEFs). In contrast, we show that B-type lamins predominantly interact with VIFs through LINC complexes to regulate cytoplasmic stiffness and contractility. We then propose a physical model mediated by the lamin–LINC complex that explains these distinct mechanical phenotypes (mechanophenotypes). To verify this model, we use dominant negative constructs and RNA interference to disrupt the LINC complexes that facilitate the interaction of the nucleus with the F-actin and VIF cytoskeletons and show that the loss of these elements results in mechanophenotypes like those observed in MEFs that lack A- or B-type lamin isoforms. Finally, we demonstrate that the loss of each lamin isoform softens the cell nucleus and enhances constricted cell migration but in turn increases migration-induced DNA damage. Together, our findings uncover distinctive roles for each of the four major lamin isoforms in maintaining nucleocytoskeletal interactions and cellular mechanics.
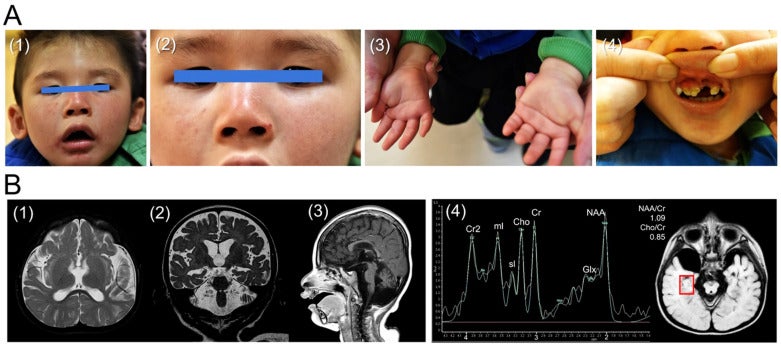
CM Lin, JH Yang, HJ Lee, YP Lin, LP Tsai, CS Hsu, GWG Luxton*, CF Hu* (2021), Whole Exome Sequencing Identifies a Novel Homozygous Missense Mutation in the CSB Protein-Encoding ERCC6 Gene in a Taiwanese Boy with Cockayne Syndrome. Life 11(11):1230. (* Equal contribution).
Background: Cockayne syndrome (CS) is a rare form of dwarfism that is characterized by progressive premature aging. CS is typically caused by mutations in the excision repair cross-complementing protein group 6 (ERCC6) gene that encodes the CS group B (CSB) protein. Using whole exome sequencing, we recently identified a novel homozygous missense mutation (Leu536Trp) in CSB in a Taiwanese boy with CS. Since the current database (Varsome) interprets this variant as likely pathogenic, we utilized a bioinformatic tool to investigate the impact of Leu536Trp as well as two other variants (Arg453Ter, Asp532Gly) in similar articles on the CSB protein structure stability. Methods: We used iterative threading assembly refinement (I-TASSER) to generate a predictive 3D structure of CSB. We calculated the change of mutation energy after residues substitution on the protein stability using I-TASSER as well as the artificial intelligence program Alphafold. Results: The Asp532Gly variant destabilized both modeled structures, while the Leu536Trp variant showed no effect on I-TASSER’s model but destabilized the Alphafold’s modeled structure. Conclusions: We propose here the first case of CS associated with a novel homozygous missense mutation (Leu536Trp) in CSB. Furthermore, we suggest that the Asp532Gly and Leu536Trp variants are both pathogenic after bioinformatic analysis of protein stability.
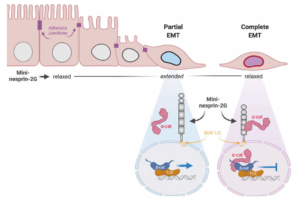
CJ Gottardi, GWG Luxton (2020), Nesprin-2G tension fine-tunes Wnt/β-catenin signaling. Journal of Cell Biology.
How LINC complexes mediate nuclear mechanotransduction remains unclear. In this issue, Déjardin, Carollo, et al. (2020. J. Cell Biol.) show that the LINC complex protein nesprin-2G is a mechanosensor of epithelial–mesenchymal transitions (EMTs), recruiting α-catenin to the nucleus to attenuate Wnt/β-catenin signaling.
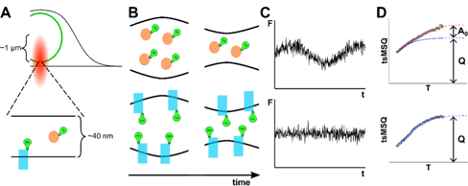
J Hennen, J Kohler, SR Karuka, Y Chen, GWG Luxton, JD Mueller (2020), Differentiating Luminal and Membrane-Associated Nuclear Envelope Proteins. Biophysical Journal.
The nuclear envelope (NE) consists of two membranes separated by the ∼40 nm wide lumen. This thin structure, being well below the resolution limit, presents a challenge to differentiating membrane-associated and luminal protein populations within the NE of living cells. Distinguishing these two populations is further complicated by the observation that diffusion time, a commonly used marker for membrane association in other cellular compartments, shows a strong dependence on molecular size within the lumen. Thus, a decrease in proteins mobility within the NE may be caused by the formation of large soluble complexes or association with the membranes. We recently observed a striking divergence in the temperature dependence of mobility for luminal and membrane-associated proteins within the NE, which provides a reliable marker for differentiating these two populations. Furthermore, previously reported nuclear membrane undulations introduce an additional fluctuation signal to luminal proteins while not affecting membrane-associated proteins. We demonstrate that this effect providing a second, independent marker distinguishing luminal and membrane-associated protein. These two independent assays for membrane-association are applied to proteins of biological interest. The luminal domain of SUN2 was previously shown to exist as a mixture of fast diffusing monomers and slowly diffusing trimers. We apply our tools to determine whether this decrease in mobility is caused solely by the three-fold increase in molecular weight or by membrane association. Finally, we use these techniques to identify the extent of membrane association for the AAA+ ATPase torsinA and explore the effect of tagging the fluorescent label to its N- or C-terminus. This work has been supported by a grant from the National Institutes of Health (R01 GM64589).
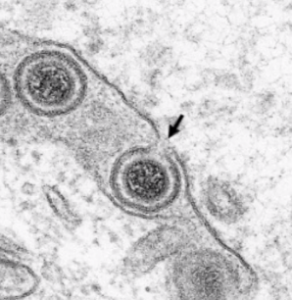
JE Hölper, BG Klupp, GWG Luxton, K Franzke, TC Mettenleiter (2020), Function of Torsin AAA+ ATPases in Pseudorabies Virus Nuclear Egress. Cells.
Newly assembled herpesvirus nucleocapsids traverse the intact nuclear envelope by a vesicle-mediated nucleo-cytoplasmic transport for final virion maturation in the cytoplasm. For this, they bud at the inner nuclear membrane resulting in primary enveloped particles in the perinuclear space (PNS) followed by fusion of the primary envelope with the outer nuclear membrane (ONM). While the conserved viral nuclear egress complex orchestrates the first steps, effectors of fusion of the primary virion envelope with the ONM are still mostly enigmatic but might include cellular proteins like SUN2 or ESCRT-III components. Here, we analyzed the influence of the only known AAA+ ATPases located in the endoplasmic reticulum and the PNS, the Torsins (Tor), on nuclear egress of the alphaherpesvirus pseudorabies virus. For this overexpression of wild type and mutant proteins as well as CRISPR/Cas9 genome editing was applied. Neither single overexpression nor gene knockout (KO) of TorA or TorB had a significant impact. However, TorA/B double KO cells showed decreased viral titers at early time points of infection and an accumulation of primary virions in the PNS pointing to a delay in capsid release during nuclear egress.
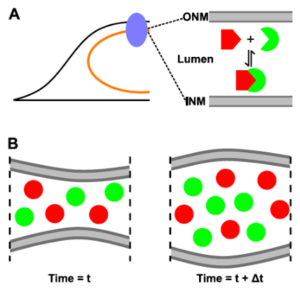
J Hennen, KH Hur, J Kohler, SR Karuka, I Angert, GWG Luxton, JD Mueller (2019), Identifying hetero-protein complexes in the nuclear envelope. Biophysical Journal.
-
Selected by the Biophysical Journal to be highlighted by a “New and Notable” article
-
-
L Lanzanò (2020), Counting the components of protein complexes in the nuclear envelope. Biophysical Journal.
-
The nucleus is delineated by the nuclear envelope (NE), which is a double membrane barrier composed of the inner and outer nuclear membranes as well as a ∼40-nm wide lumen. In addition to its barrier function, the NE acts as a critical signaling node for a variety of cellular processes, which are mediated by protein complexes within this subcellular compartment. Although fluorescence fluctuation spectroscopy is a powerful tool for characterizing protein complexes in living cells, it was recently demonstrated that conventional fluorescence fluctuation spectroscopy methods are not suitable for applications in the NE because of the presence of slow nuclear membrane undulations. We previously addressed this challenge by developing time-shifted mean-segmented Q (tsMSQ) analysis and applied it to successfully characterize protein homo-oligomerization in the NE. However, many NE complexes, such as the linker of the nucleoskeleton and cytoskeleton complex, are formed by heterotypic interactions, which single-color tsMSQ is unable to characterize. Here, we describe the development of dual-color (DC) tsMSQ to analyze NE heteroprotein complexes built from proteins that carry two spectrally distinct fluorescent labels. Experiments performed on model systems demonstrate that DC tsMSQ properly identifies heteroprotein complexes and their stoichiometry in the NE by accounting for spectral cross talk and local volume fluctuations. Finally, we applied DC tsMSQ to study the assembly of the linker of the nucleoskeleton and cytoskeleton complex, a heteroprotein complex composed of Klarsicht/ANC-1/SYNE homology and Sad1/UNC-84 (SUN) proteins, in the NE of living cells. Using DC tsMSQ, we demonstrate the ability of the SUN protein SUN2 and the Klarsicht/ANC-1/SYNE homology protein nesprin-2 to form a heterocomplex in vivo. Our results are consistent with previously published in vitro studies and demonstrate the utility of the DC tsMSQ technique for characterizing NE heteroprotein complexes.
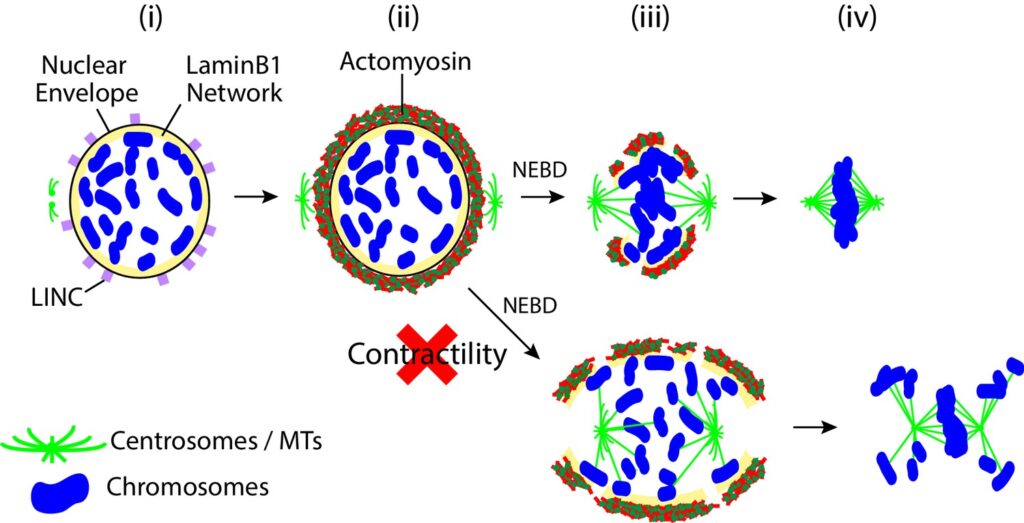
AJR Booth, Z Yue, JK Eykelenboom, T Stiff, GWG Luxton, H Hochegger, TU Tanaka (2019), Contractile acto-myosin network on nuclear envelope remnants positions human chromosomes for mitosis. eLife.
To ensure proper segregation during mitosis, chromosomes must be efficiently captured by spindle microtubules and subsequently aligned on the mitotic spindle. The efficacy of chromosome interaction with the spindle can be influenced by how widely chromosomes are scattered in space. Here, we quantify chromosome-scattering volume (CSV) and find that it is reduced soon after nuclear envelope breakdown (NEBD) in human cells. The CSV reduction occurs primarily independently of microtubules and is therefore not an outcome of interactions between chromosomes and the spindle. We find that, prior to NEBD, an acto-myosin network is assembled in a LINC complex-dependent manner on the cytoplasmic surface of the nuclear envelope. This acto-myosin network remains on nuclear envelope remnants soon after NEBD, and its myosin-II-mediated contraction reduces CSV and facilitates timely chromosome congression and correct segregation. Thus, we find a novel mechanism that positions chromosomes in early mitosis to ensure efficient and correct chromosome–spindle interactions.
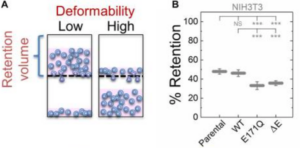
NK Gill, C Ly, PH Kim, CA Saunders, LG Fong, SG Young, GWG Luxton*, AC Rowat* (2019), DYT1 Dystonia Patient-Derived Fibroblasts Have Increased Deformability and Susceptibility to Damage by Mechanical Forces. Frontiers in Cellular and Developmental Biology. (*Equal Contribution)
DYT1 dystonia is a neurological movement disorder that is caused by a loss-of-function mutation in the DYT1/TOR1A gene, which encodes torsinA, a conserved luminal ATPases-associated with various cellular activities (AAA+) protein. TorsinA is required for the assembly of functional linker of nucleoskeleton and cytoskeleton (LINC) complexes, and consequently the mechanical integration of the nucleus and the cytoskeleton. Despite the potential implications of altered mechanobiology in dystonia pathogenesis, the role of torsinA in regulating cellular mechanical phenotype, or mechanotype, in DYT1 dystonia remains unknown. Here, we define the deformability of mouse fibroblasts lacking functional torsinA as well as human fibroblasts isolated from DYT1 dystonia patients. We find that the deletion of torsinA or the expression of torsinA containing the DYT1 dystonia-causing ΔE302/303 (ΔE) mutation results in more deformable cells. We observe a similar increased deformability of mouse fibroblasts that lack lamina-associated polypeptide 1 (LAP1), which interacts with and stimulates the ATPase activity of torsinA in vitro, as well as with the absence of the LINC complex proteins, Sad1/UNC-84 1 (SUN1) and SUN2, lamin A/C, or lamin B1. Consistent with these findings, we also determine that DYT1 dystonia patient-derived fibroblasts are more compliant than fibroblasts isolated from unafflicted individuals. DYT1 dystonia patient-derived fibroblasts also exhibit increased nuclear strain and decreased viability following mechanical stretch. Taken together, our results establish the foundation for future mechanistic studies of the role of cellular mechanotype and LINC-dependent nuclear-cytoskeletal coupling in regulating cell survival following exposure to mechanical stresses.
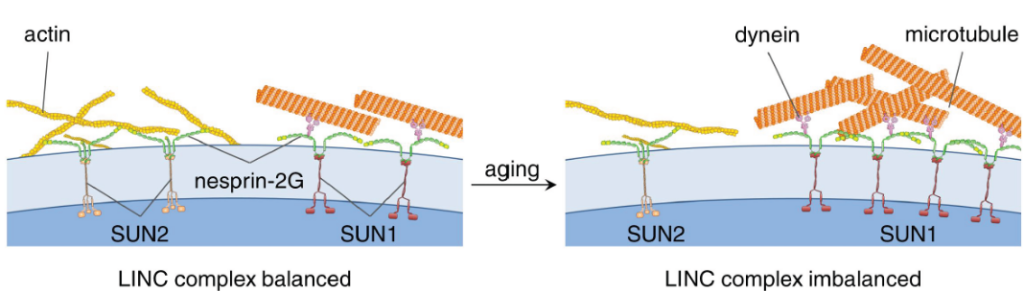
W Chang, Y Wang, GWG Luxton, C Östlund, HJ Worman, GG Gundersen (2019), Imbalanced nucleocytoskeletal connections create common polarity defects in progeria and physiological aging. Proceedings of the National Academy of Sciences.
Studies of the accelerated aging disorder Hutchinson–Gilford progeria syndrome (HGPS) can potentially reveal cellular defects associated with physiological aging. HGPS results from expression and abnormal nuclear envelope association of a farnesylated, truncated variant of prelamin A called “progerin.” We surveyed the diffusional mobilities of nuclear membrane proteins to identify proximal effects of progerin expression. The mobilities of three proteins—SUN2, nesprin-2G, and emerin—were reduced in fibroblasts from children with HGPS compared with those in normal fibroblasts. These proteins function together in nuclear movement and centrosome orientation in fibroblasts polarizing for migration. Both processes were impaired in fibroblasts from children with HGPS and in NIH 3T3 fibroblasts expressing progerin, but were restored by inhibiting protein farnesylation. Progerin affected both the coupling of the nucleus to actin cables and the oriented flow of the cables necessary for nuclear movement and centrosome orientation. Progerin overexpression increased levels of SUN1, which couples the nucleus to microtubules through nesprin-2G and dynein, and microtubule association with the nucleus. Reducing microtubule-nuclear connections through SUN1 depletion or dynein inhibition rescued the polarity defects. Nuclear movement and centrosome orientation were also defective in fibroblasts from normal individuals over 60 y, and both defects were rescued by reducing the increased level of SUN1 in these cells or inhibiting dynein. Our results identify imbalanced nuclear engagement of the cytoskeleton (microtubules: high; actin filaments: low) as the basis for intrinsic cell polarity defects in HGPS and physiological aging and suggest that rebalancing the connections can ameliorate the defects.
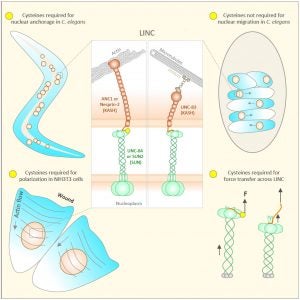
Current Biology.
Many nuclear positioning events involve linker of nucleoskeleton and cytoskeleton (LINC) complexes, which transmit forces generated by the cytoskeleton across the nuclear envelope. LINC complexes are formed by trans-luminal interactions between inner nuclear membrane SUN proteins and outer nuclear membrane KASH proteins, but how these interactions are regulated is poorly understood. We combine in vivo C. elegans genetics, in vitro wounded fibroblast polarization, and in silico molecular dynamic simulations to elucidate mechanisms of LINC complexes. The extension of the KASH domain by a single alanine residue or the mutation of the conserved tyrosine at -7 completely blocked the nuclear migration function of C. elegans UNC-83. Analogous mutations at -7 of mouse nesprin-2 disrupted rearward nuclear movements in NIH3T3 cells, but did not disrupt ANC-1 in nuclear anchorage. Furthermore, conserved cysteines predicted to form a disulfide bond between SUN and KASH proteins are important for the function of certain LINC complexes and might promote a developmental switch between nuclear migration and nuclear anchorage. Mutations of conserved cysteines in SUN or KASH disrupted ANC-1 dependent nuclear anchorage in C. elegans and Nesprin-2G dependent nuclear movements in polarizing fibroblasts. However, the SUN cysteine mutation did not disrupt nuclear migration. Moreover, molecular dynamic simulations showed that a disulfide bond is necessary for the maximal transmission of cytoskeleton-generated forces by LINC complexes in silico. Thus, we have demonstrated functions for SUN-KASH binding interfaces, including a predicted intermolecular disulfide bond, as mechanistic determinants of nuclear positioning and may represent targets for regulation.
Methods.
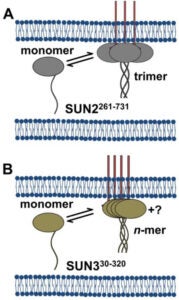
Analysis of fluorescence fluctuation experiments by the mean-segmented Q (MSQ) method was recently used to successfully characterize the oligomeric state and mobility of proteins within the nuclear envelope (NE) of living cells. However, two significant shortcomings of MSQ were recognized. Non-ideal detector behavior due to dead-time and afterpulsing as well as the lack of error analysis currently limit the potential of MSQ. This paper presents time-shifted MSQ (tsMSQ), a new formulation of MSQ that is robust with respect to dead-time and afterpulsing. In addition, a protocol for performing error analysis on tsMSQ data is introduced to assess the quality of fit models and estimate the uncertainties of fit parameters. Together, these developments significantly simplify and improve the analysis of fluorescence fluctuation data taken within the NE. To demonstrate these new developments, tsMSQ was used to characterize the oligomeric state and mobility of the luminal domains of two inner nuclear membrane SUN proteins. The results for the luminal domain of SUN2 obtained through tsMSQ without correction for non-ideal detector effects agree with a recent study that was conducted using the original MSQ formulation. Finally, tsMSQ was applied to characterize the oligomeric state and mobility of the luminal domain of the germline-restricted SUN3.
. (*Equal Contribution)
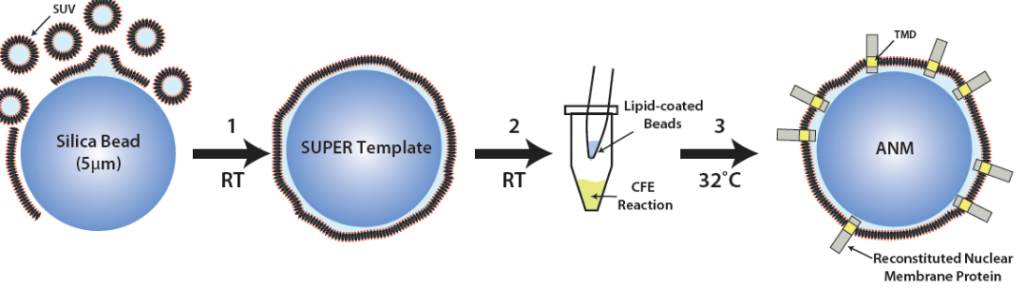
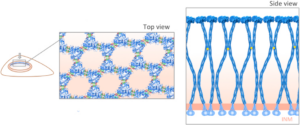
The linker of nucleoskeleton and cytoskeleton (LINC) is a conserved nuclear envelope-spanning molecular bridge that is responsible for the mechanical integration of the nucleus with the cytoskeleton. LINC complexes are formed by a transluminal interaction between the outer and inner nuclear membrane KASH and SUN proteins, respectively. Despite recent structural insights, our mechanistic understanding of LINC complex assembly remains limited by the lack of an experimental system for its in vitro reconstitution and manipulation. Here, we describe artificial nuclear membranes (ANMs) as a synthetic biology platform based on mammalian cell-free expression for the rapid reconstitution of SUN proteins in supported lipid bilayers. We demonstrate that SUN1 and SUN2 are oriented in ANMs with solvent-exposed C-terminal KASH-binding SUN domains. We also find that SUN2 possesses a single transmembrane domain, while SUN1 possesses three. Finally, SUN protein-containing ANMs bind synthetic KASH peptides, thereby reconstituting the LINC complex core. This work represents the first in vitro reconstitution of KASH-binding SUN proteins in supported lipid bilayers using cell-free expression, which will be invaluable for testing proposed models of LINC complex assembly and its regulation.
. (*Equal Contribution)
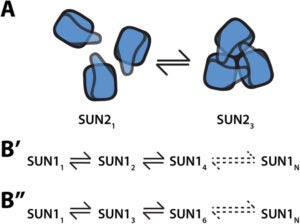
Linker-of-nucleoskeleton-and-cytoskeleton (LINC) complexes are conserved molecular bridges within the nuclear envelope that mediate mechanical force transmission into the nucleoplasm. The core of a LINC complex is formed by a transluminal interaction between the outer and inner nuclear membrane KASH and SUN proteins, respectively. Mammals encode six KASH proteins and five SUN proteins. Recently, KASH proteins were shown to bind to the domain interfaces of trimeric SUN2 proteins in vitro. However, neither the existence of SUN2 trimers in living cells nor the extent to which other SUN proteins conform to this assembly state have been tested experimentally. Here we extend the application of fluorescence fluctuation spectroscopy to quantify SUN protein oligomerization in the nuclear envelopes of living cells. Using this approach, we demonstrate for the first time that SUN2 trimerizes in vivo and we demonstrate that the in vivo oligomerization of SUN1 is not limited to a trimer. In addition, we provide evidence to support the existence of potential regulators of SUN protein oligomerization in the nuclear envelope. The differential SUN protein oligomerization illustrated here suggests that SUN proteins may have evolved to form different assembly states in order to participate in diverse mechanotransduction events.
.
The LINC complex is found in a wide variety of organisms and is formed by the transluminal interaction between outer- and inner-nuclear-membrane KASH and SUN proteins, respectively. Most extensively studied are SUN1 and SUN2 proteins, which are widely expressed in mammals. Although SUN1 and SUN2 play functionally redundant roles in several cellular processes, more recent studies have revealed diverse and distinct functions for SUN1. While several recent in vitro structural studies have revealed the molecular details of various fragments of SUN2, no such structural information is available for SUN1. Herein, we conduct a systematic analysis of the molecular relationships between SUN1 and SUN2, highlighting key similarities and differences that could lead to clues into their distinct functions. We use a wide range of computational tools, including multiple sequence alignments, homology modeling, molecular docking, and molecular dynamic simulations, to predict structural differences between SUN1 and SUN2, with the goal of understanding the molecular mechanisms underlying SUN1 oligomerization in the nuclear envelope. Our simulations suggest that the structural model of SUN1 is stable in a trimeric state and that SUN1 trimers can associate through their SUN domains to form lateral complexes. We also ask whether SUN1 could adopt an inactive monomeric conformation as seen in SUN2. Our results imply that the KASH binding domain of SUN1 is also inhibited in monomeric SUN1 but through weaker interactions than in monomeric SUN2.
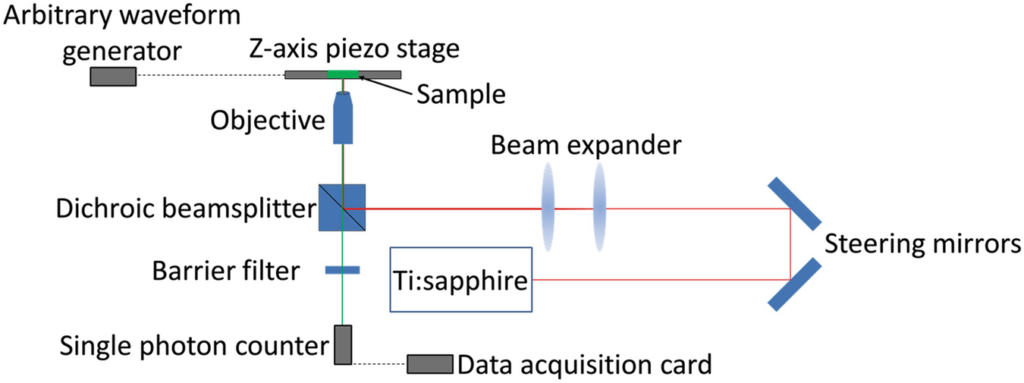
. Methods in Molecular Biology. (*Equal Contribution)
Linkers of nucleoskeleton and cytoskeleton (LINC) complexes are conserved nuclear envelope (NE) spanning molecular bridges which mechanically integrate the nucleus with the cytoskeleton and mediate force transmission into the nucleoplasm. Despite their critical roles in fundamental cellular processes such as meiotic chromosome and nuclear positioning, the mechanism of LINC complex assembly in cells remains unclear. To begin to address this deficit, we recently developed z-scan fluorescence fluctuation spectroscopy (FFS) and brightness analysis as a method for quantifying the oligomeric states of fluorescent protein-tagged NE proteins including nesprins and SUN proteins. Since the homo-oligomerization of SUN2 is critical for its ability to interact with nesprins within the perinuclear space, the knowledge obtained through quantitative brightness experiments reveals important insights into the in vivo mechanisms of LINC complex assembly. Here we describe the procedure we use to determine the brightness of proteins in the NE of living cells. In addition to the measurement procedure, we discuss the instrumentation requirements and present the results of applying this procedure to measure the brightness of nesprin-2 and SUN2.
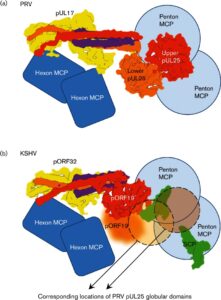
.
Inside the virions of α-herpesviruses, tegument protein pUL25 anchors the tegument to capsid vertices through direct interactions with tegument proteins pUL17 and pUL36. In addition to promoting virion assembly, both pUL25 and pUL36 are critical for intracellular microtubule-dependent capsid transport. Despite these essential roles during infection, the stoichiometry and precise organization of pUL25 and pUL36 on the capsid surface remain controversial due to the insufficient resolution of existing reconstructions from cryo-electron microscopy (cryoEM). Here, we report a three-dimensional (3D) icosahedral reconstruction of pseudorabies virus (PRV), a varicellovirus of the α-herpesvirinae subfamily, obtained by electron-counting cryoEM at 4.9 Å resolution. Our reconstruction resolves a dimer of pUL25 forming a capsid-associated tegument complex with pUL36 and pUL17 through a coiled coil helix bundle, thus correcting previous misinterpretations. A comparison between reconstructions of PRV and the γ-herpesvirus Kaposi’s sarcoma-associated herpesvirus (KSHV) reinforces their similar architectures and establishes important subfamily differences in the capsid-tegument interface.
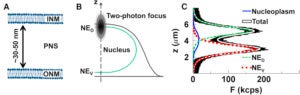
.
Brightness analysis of fluorescence fluctuation experiments has been used to successfully measure the oligomeric state of proteins at the plasma membrane, in the nucleoplasm, and in the cytoplasm of living cells. Here we extend brightness analysis to the nuclear envelope (NE), a double membrane barrier separating the cytoplasm from the nucleoplasm. Results obtained by applying conventional brightness analysis to fluorescently tagged proteins within the NE exhibited an unusual concentration dependence. Similarly, the autocorrelation function of the fluorescence fluctuations exhibited unexpected changes with protein concentration. These observations motivated the application of mean-segmented Q analysis, which identified the existence of a fluctuation process distinct from molecular diffusion in the NE. We propose that small changes in the separation of the inner and outer nuclear membrane are responsible for the additional fluctuation process, as suggested by results obtained for luminal and nuclear membrane-associated EGFP-tagged proteins. Finally, we applied these insights to study the oligomerization of the luminal domains of two nuclear membrane proteins, nesprin-2 and SUN2, which interact transluminally to form a nuclear envelope-spanning linker molecular bridge known as the linker of the nucleoskeleton and cytoskeleton complex.
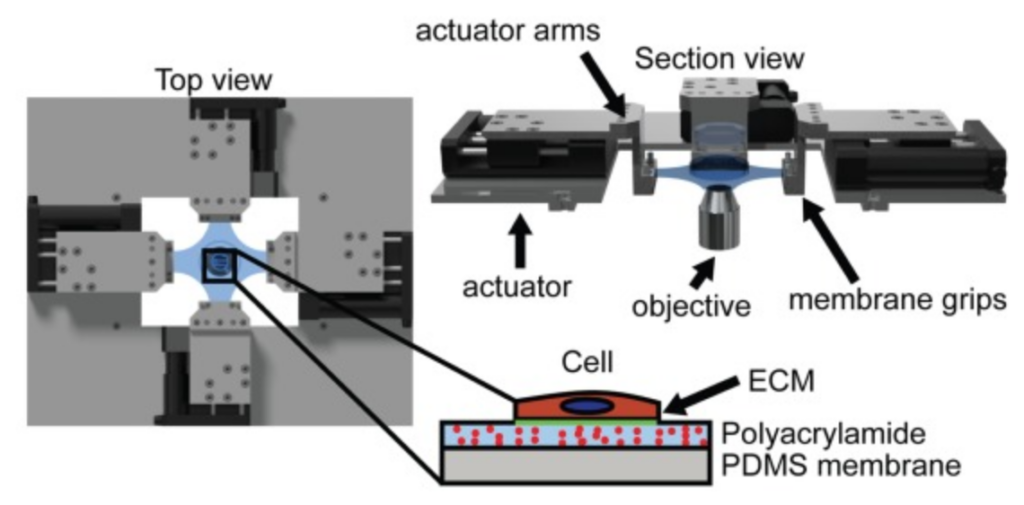
.
The stress in a cell due to extracellular mechanical stimulus is determined by its mechanical properties, and the structural organization of many adherent cells suggests that their properties are anisotropic. This anisotropy may significantly influence the cells’ mechanotransductive response to complex loads, and has important implications for development of accurate models of tissue biomechanics. Standard methods for measuring cellular mechanics report linear moduli that cannot capture large-deformation anisotropic properties, which in a continuum mechanics framework are best described by a strain energy density function (SED). In tissues, the SED is most robustly measured using biaxial testing. Here, we describe a cellular microbiaxial stretching (CμBS) method that modifies this tissue-scale approach to measure the anisotropic elastic behavior of individual vascular smooth muscle cells (VSMCs) with nativelike cytoarchitecture. Using CμBS, we reveal that VSMCs are highly anisotropic under large deformations. We then characterize a Holzapfel-Gasser-Ogden type SED for individual VSMCs and find that architecture-dependent properties of the cells can be robustly described using a formulation solely based on the organization of their actin cytoskeleton. These results suggest that cellular anisotropy should be considered when developing biomechanical models, and could play an important role in cellular mechano-adaptation.
. (*Equal Contribution)
-
Highlighted on the cover of the journal
-
Selected by the Journal of Cell Biology to be highlighted by a “Spotlight” article
-
DA Starr, LS Rose (2017), TorsinA regulates the LINC to moving nuclei. Journal of Cell Biology.
-
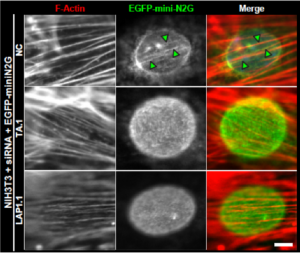
The nucleus is positioned toward the rear of most migratory cells. In fibroblasts and myoblasts polarizing for migration, retrograde actin flow moves the nucleus rearward, resulting in the orientation of the centrosome in the direction of migration. In this study, we report that the nuclear envelope-localized AAA+ (ATPase associated with various cellular activities) torsinA (TA) and its activator, the inner nuclear membrane protein lamina-associated polypeptide 1 (LAP1), are required for rearward nuclear movement during centrosome orientation in migrating fibroblasts. Both TA and LAP1 contributed to the assembly of transmembrane actin-associated nuclear (TAN) lines, which couple the nucleus to dorsal perinuclear actin cables undergoing retrograde flow. In addition, TA localized to TAN lines and was necessary for the proper mobility of EGFP-mini-nesprin-2G, a functional TAN line reporter construct, within the nuclear envelope. Furthermore, TA and LAP1 were indispensable for the retrograde flow of dorsal perinuclear actin cables, supporting the recently proposed function for the nucleus in spatially organizing actin flow and cytoplasmic polarity. Collectively, these results identify TA as a key regulator of actin-dependent rearward nuclear movement during centrosome orientation.
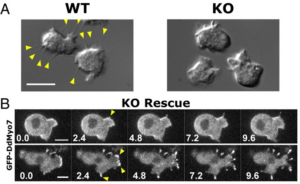
The formation of filopodia in Metazoa and Amoebozoa requires the activity of myosin 10 (Myo10) in mammalian cells and of Dictyostelium unconventional myosin 7 (DdMyo7) in the social amoeba Dictyostelium. However, the exact roles of these MyTH4-FERM myosins (myosin tail homology 4-band 4.1, ezrin, radixin, moesin; MF) in the initiation and elongation of filopodia are not well defined and may reflect conserved functions among phylogenetically diverse MF myosins. Phylogenetic analysis of MF myosin domains suggests that a single ancestral MF myosin existed with a structure similar to DdMyo7, which has two MF domains, and that subsequent duplications in the metazoan lineage produced its functional homolog Myo10. The essential functional features of the DdMyo7 myosin were identified using quantitative live-cell imaging to characterize the ability of various mutants to rescue filopod formation in myo7-null cells. The two MF domains were found to function redundantly in filopod formation with the C-terminal FERM domain regulating both the number of filopodia and their elongation velocity. DdMyo7 mutants consisting solely of the motor plus a single MyTH4 domain were found to be capable of rescuing the formation of filopodia, establishing the minimal elements necessary for the function of this myosin. Interestingly, a chimeric myosin with the Myo10 MF domain fused to the DdMyo7 motor also was capable of rescuing filopod formation in the myo7-null mutant, supporting fundamental functional conservation between these two distant myosins. Together, these findings reveal that MF myosins have an ancient and conserved role in filopod formation.
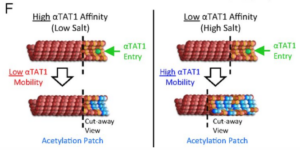
Microtubules are structural polymers inside of cells that are subject to posttranslational modifications. These posttranslational modifications create functionally distinct subsets of microtubule networks in the cell, and acetylation is the only modification that takes place in the hollow lumen of the microtubule. Although it is known that the α-tubulin acetyltransferase (αTAT1) is the primary enzyme responsible for microtubule acetylation, the mechanism for how αTAT1 enters the microtubule lumen to access its acetylation sites is not well understood. By performing biochemical assays, fluorescence and electron microscopy experiments, and computational simulations, we found that αTAT1 enters the microtubule lumen through the microtubule ends, and through bends or breaks in the lattice. Thus, microtubule structure is an important determinant in the acetylation process. In addition, once αTAT1 enters the microtubule lumen, the mobility of αTAT1 within the lumen is controlled by the affinity of αTAT1 for its acetylation sites, due to the rapid rebinding of αTAT1 onto highly concentrated α-tubulin acetylation sites. These results have important implications for how acetylation could gradually accumulate on stable subsets of microtubules inside of the cell.
The view of the cell nucleus has evolved from an isolated, static organelle to a dynamic structure integrated with other mechanical elements of the cell. Both dynamics and integration appear to contribute to a mechanical regulation of genome expression. Here, we review physical structures inside the nucleus at different length scales and the dynamic reorganization modulated by cellular forces. First, we discuss nuclear organization focusing on self-assembly and disassembly of DNA structures and various nuclear bodies. We then discuss the importance of connections from the chromatin fiber through the nuclear envelope to the rest of the cell as they relate to mechanobiology. Finally, we discuss how cell stimulation, both chemical and physical, can alter nuclear structures and ultimately cellular function in healthy cells and in some model diseases. The view of chromatin and nuclear bodies mechanical entities integrated with force generation from the cytoskeleton combines polymer physics with cell biology and medicine.
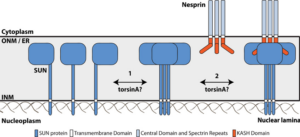
Mechanical forces generated by nuclear-cytoskeletal coupling through the LINC (linker of nucleoskeleton and cytoskeleton) complex, an evolutionarily conserved molecular bridge in the nuclear envelope (NE), are critical for the execution of wholesale nuclear positioning events in migrating and dividing cells, chromosome dynamics during meiosis, and mechanotransduction. LINC complexes consist of outer (KASH (Klarsicht, ANC-1, and Syne homology)) and inner (SUN (Sad1, UNC-84)) nuclear membrane proteins. KASH proteins interact with the cytoskeleton in the cytoplasm and SUN proteins in the perinuclear space of the NE. In the nucleoplasm, SUN proteins interact with A-type nuclear lamins and chromatin-binding proteins. Recent structural insights into the KASH-SUN interaction have generated several questions regarding how LINC complex assembly and function might be regulated within the perinuclear space. Here we discuss potential LINC regulatory mechanisms and focus on the potential role of AAA+ (ATPases associated with various cellular activities) protein, torsinA, as a LINC complex regulator within the NE. We also examine how defects in LINC complex regulation by torsinA may contribute to the pathogenesis of the human neurological movement disorder, DYT1 dystonia.
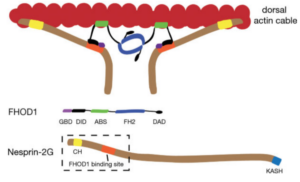
Active positioning of the nucleus is integral to division, migration, and differentiation of mammalian cells. Fibroblasts polarizing for migration orient their centrosomes by actin-dependent nuclear movement. This nuclear movement depends on nesprin-2 giant (N2G), a large, actin-binding outer nuclear membrane component of transmembrane actin-associated (TAN) lines that couple nuclei to moving actin cables. Here, we identify the diaphanous formin FHOD1 as an interaction partner of N2G. Silencing FHOD1 expression or expression of fragments containing binding sites of N2G or FHOD1 disrupted nuclear movement and centrosome orientation in polarizing fibroblasts. Unexpectedly, silencing of FHOD1 expression did not affect the formation or rearward flow of dorsal actin cables required for nuclear positioning. Rather, N2G-FHOD1 interaction provided a second connection to actin cables essential for TAN line formation and thus nuclear movement. These results reveal a unique function for a formin in coupling an organelle to actin filaments for translocation and suggest that TAN lines require multi-point attachments to actin cables to resist the large forces necessary to move the nucleus.
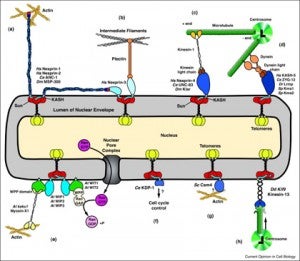
Nuclear-cytoskeletal connections are central to fundamental cellular processes, including nuclear positioning and chromosome movements in meiosis. The cytoskeleton is coupled to the nucleoskeleton through conserved KASH-SUN bridges, or LINC complexes, that span the nuclear envelope. KASH proteins localize to the outer nuclear membrane where they connect the nucleus to the cytoskeleton. New findings have expanded the functional diversity of KASH proteins, showing that they interact with microtubule motors, actin, intermediate filaments, a nonconventional myosin, RanGAP, and each other. The role of KASH proteins in cellular mechanics is discussed. Genetic mutations in KASH proteins are associated with autism, hearing loss, cancer, muscular dystrophy and other diseases.
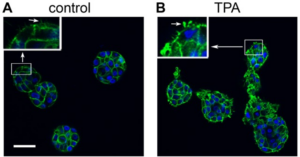
The potent tumor promoter 12-O-tetradecanoylphorbol-13-acetate (TPA) alters many cellular processes through activation of its receptor protein kinase C (PKC), including gene expression, cell cycle, and the regulation of cell morphology, raising an important question for developing targeted methods to prevent cancer: which effects of TPA are crucial for carcinogenesis? To address this question, we studied TPA action in the 3-dimensional (3D) MCF10A human breast epithelial cell system, which models important features of in vivo epithelial tissue including growth constraints, structural organization of cells, and establishment of a basement membrane. MCF10A cells, which are immortalized but nontumorigenic, form hollow, spheroid structures in 3D culture referred to as acini. The development of normal acini requires the tight spatiotemporal regulation of cellular proliferation, polarization, apoptosis, and growth arrest. Treatment of MCF10A acini with TPA caused the appearance of multi-acinar structures. Surprisingly, this phenotype did not involve an increase in cell number or major changes in cell death, and polarization. Instead, live cell and confocal microscopy revealed that TPA stimulates MCF10A acini to aggregate. TPA induces the PKC-dependent production of actin-based protrusions, which leads to the formation of cellular bridges between acini, the clustering of acini, and allows cells to move into adjacent acini. During this process, the integrity of the laminin V basement membrane is disrupted, while E-cadherin-based cell-cell contacts remain intact. Altogether, our results show that under the biochemical and structural constraints of epithelial tissue, as modeled by the 3D MCF10A system, TPA induces a novel PKC-dependent phenotype that resembles local invasion. Of the many effects caused by TPA, these studies highlight the aggressive production of actin-based cellular protrusions as a potentially important event along the pathway to carcinogenesis.
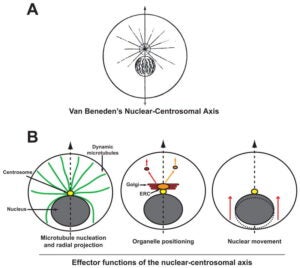
GWG Luxton, GG Gundersen (2011)
A hallmark of polarity in most migrating cells is the orientation of the nuclear centrosomal (NC) axis relative to the front-back cellular axis. Here, we review ‘effector functions’ associated with the NC axis during cell migration. We highlight recent research that has demonstrated that the orientation of the NC axis depends upon the coordinated, but separate positioning of the nucleus and the centrosome. We stress the importance of environmental factors such as cell-cell contacts and substrate topology for NC axis orientation. Finally, we summarize tests of the significance of this axis for cell migration and disease.
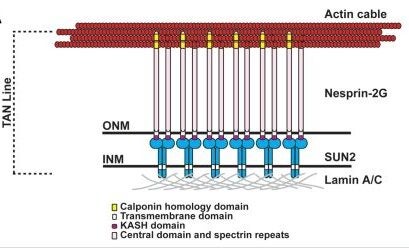
Nuclear position is actively controlled and can be adjusted according to the needs of a cell by nuclear movement. Microtubules mediate the majority of nuclear movements studied to date, although examples of nuclear movements mediated by the actin cytoskeleton have been described. One such actin-dependent nuclear movement occurs during centrosome orientation in fibroblasts polarizing for migration. Here, the centrosome is maintained at the cell center while the nucleus is moved to the cell rear by actin retrograde flow thus positioning the centrosome between the nucleus and the leading edge of the cell. We have explored the molecular mechanism for actin dependent movement of the nucleus during centrosome centration. We found that a novel linear array of nuclear envelope membrane proteins composed of nesprin-2G and SUN2, called transmembrane actin-associated nuclear (TAN) lines, couple the nucleus to moving actin cables resulting in the nucleus being positioned toward the cell rear. TAN lines are anchored by A-type lamins and this allows the forces generated by the actin cytoskeleton to be transmitted across the nuclear envelope to move the nucleus. Here we review the data supporting this mechanism for nuclear movement, discuss questions remaining to be addressed and consider how this new mechanism of nuclear movement may shed light on human disease.
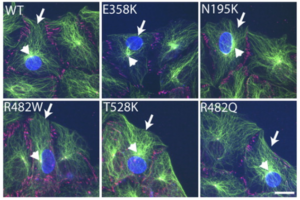
Mutations in LMNA, which encodes A-type lamins, result in disparate diseases, known collectively as laminopathies, that affect distinct tissues, including striated muscle and adipose tissue. Lamins provide structural support for the nucleus and sites of attachment for chromatin, and defects in these functions may contribute to disease pathogenesis. Recent studies suggest that A-type lamins may facilitate connections between the nucleus and the cytoskeleton mediated by nuclear envelope nesprin and SUN proteins. In mammalian cells, however, interfering with A-type lamins does not affect the localization of these proteins. Here, we used centrosome orientation in fibroblasts, which requires separate nuclear and centrosome positioning pathways, as a model system to understand how LMNA mutations affect nucleus-cytoskeletal connections. We find that LMNA mutations causing striated muscle diseases block actin-dependent nuclear movement, whereas most that affect adipose tissue inhibit microtubule-dependent centrosome positioning. Genetic deletion or transient depletion of A-type lamins also blocked nuclear movement, showing that mutations affecting muscle exhibit the null phenotype. Lack of A-type lamins, or expression of variants that cause striated muscle disease, did not affect assembly of nesprin-2G and SUN2 into transmembrane actin-associated nuclear (TAN) lines that attach the nucleus to retrogradely moving actin cables. Nesprin-2G TAN lines were less stable, however, and slipped over the nucleus rather than moving with it, indicating that they were not anchored. Nesprin-2G TAN lines also slipped in SUN2-depleted cells. Our results establish A-type lamins as anchors for nesprin-2G–SUN2 TAN lines to allow productive movement and proper positioning of the nucleus by actin.
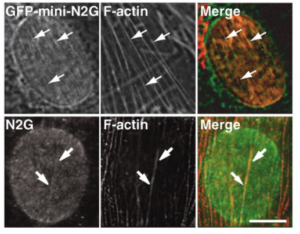
Nuclei move to specific locations to polarize migrating and differentiating cells. Many nuclear movements are microtubule-dependent. However, nuclear movement to reorient the centrosome in migrating fibroblasts occurs through an unknown actin-dependent mechanism. We found that linear arrays of outer (nesprin2G) and inner (SUN2) nuclear membrane proteins assembled on and moved with retrogradely moving dorsal actin cables during nuclear movement in polarizing fibroblasts. Inhibition of nesprin2G, SUN2, or actin prevented nuclear movement and centrosome reorientation. The coupling of actin cables to the nuclear membrane for nuclear movement via specific membrane proteins indicates that, like plasma membrane integrins, nuclear membrane proteins assemble into actin-dependent arrays for force transduction.
GWG Luxton, GG Gundersen (2007),
The reversible acetylation of lysine residues is an important posttranslational modification for the regulation of histones, transcription factors, chaperones, and microtubules. In a recent article in Molecular Cell, Zhang et al. (2007) describe a new target of reversible acetylation, the actin binding protein cortactin.
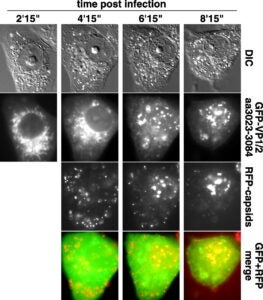
The herpesvirus tegument is a layer of viral and cellular proteins located between the capsid and envelope of the virion. The VP1/2 tegument protein is critical for the propagation of all herpesviruses examined. Using an infectious clone of the alphaherpesvirus pseudorabies virus, we have made a collection of truncation and in-frame deletion mutations within the VP1/2 gene (UL36) and examined the resulting viruses for spread between cells. We found that the majority of the VP1/2 protein either was essential for virus propagation or did not tolerate large deletions. A recently described amino-terminal deubiquitinase-encoding domain was dispensable for alphaherpesvirus propagation, but the rate of propagation in an epithelial cell line and the frequency of transport in axons of primary sensory neurons were both reduced. We mapped one essential domain to a conserved sequence at the VP1/2 carboxy terminus and demonstrated that this domain sufficient to redirect the green fluorescent protein to capsid assemblons in nuclei of infected cells.
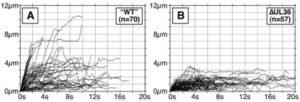
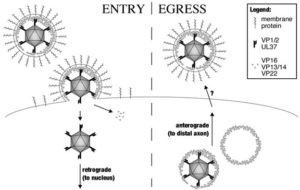
Transport of capsids in cells is critical to alphaherpesvirus infection and pathogenesis; however, viral factors required for transport have yet to be identified. Here we provide a detailed examination of capsid dynamics during the egress phase of infection in Vero cells infected with pseudorabies virus. We demonstrate that the VP1/2 tegument protein is required for processive microtubule-based transport of capsids in the cytoplasm. A second tegument protein that binds to VP1/2, UL37, was necessary for wild-type transport but was not essential for this process. Both proteins were also required for efficient nuclear egress of capsids to the cytoplasm.
The American trypanosome, Trypanosoma cruzi, can invade non‐phagocytic cell types by a G‐protein‐mediated, calcium‐dependent mechanism, in which the cell’s natural puncture repair mechanism is usurped in order to recruit lysosomes to the parasite/host cell junction or ‘parasite synapse.’ The fusion of lysosomes necessary for construction of the nascent parasitophorous vacuole is achieved by directed trafficking along microtubules. We demonstrate altered host cell microtubule dynamics during the initial stages of the entry process involving de novo microtubule polymerization from the cytoplasmic face of the parasite synapse which appears to serve as a secondary microtubule organizing centre. The net result of these dynamic changes to the host cell’s microtubule cytoskeleton is the development of the necessary infrastructure for transport of lysosomes to the parasite synapse.
-
Highlighted on the cover of the journal
-
Selected by Proc. Natl. Acad. Sci. U.S.A. to be highlighted by a “New research in” article:
-
UF Greber (2005), Viral trafficking violations in axons: The herpesvirus case.
-
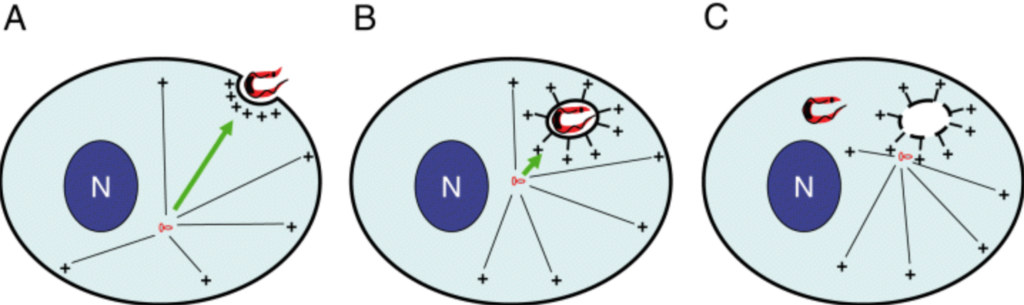
The capsids of neurotropic herpesviruses have the remarkable ability to move in specific directions within axons. By modulating bidirectional capsid transport to favor either retrograde (minus-end) or anterograde (plus-end) motion, these viruses travel to sensory ganglia or peripheral tissue at specific stages of infection. By using correlative motion analysis to simultaneously monitor the trafficking of distinct viral proteins in living neurons, we demonstrate that viral “tegument” proteins are complexed to capsids moving in axons. The removal of a subset of tegument proteins from capsids invariably preceded retrograde transport to the cell body in sensory ganglia, whereas addition of these proteins was coupled to anterograde transport of progeny capsids to the distal axon. Although capsid transport never occurred without associated tegument proteins, anterograde-specific tegument proteins were competent to travel to the distal axon independent of capsids. These findings are compatible with a model of viral bidirectional transport in which tegument proteins direct capsid traffic to specific intracellular locations during the infectious cycle.
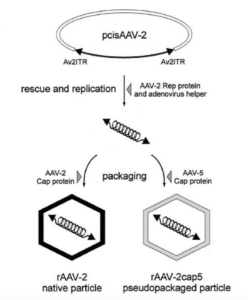
In the presence of complementing adeno-associated virus type 2 (AAV-2) Rep proteins, AAV-2 genomes can be pseudotyped with the AAV-5 capsid to assemble infectious virions. Using this pseudotyping strategy, the involvement of the ubiquitin-proteasome system in AAV-5 and AAV-2 capsid-mediated infections was compared. A recombinant AAV-2 (rAAV-2) proviral luciferase construct was packaged into both AAV-2 and AAV-5 capsid particles, and transduction efficiencies in a number of cell lines were compared. Using luciferase expression as the end point, we demonstrated that coadministration of the viruses with proteasome inhibitors not only increased the transduction efficiency of rAAV-2, as previously reported, but also augmented rAAV-5-mediated gene transfer. Increased transgene expression was independent of viral genome stability, since there was no significant difference in the amounts of internalized viral DNA in the presence or absence of proteasome inhibitors. Western blot assays of immunoprecipitated viral capsid proteins from infected HeLa cell lysates and in vitro reconstitution experiments revealed evidence for ubiquitin conjugation of both AAV-2 and AAV-5 capsids. Interestingly, heat-denatured virus particles were preferential substrates for in vitro ubiquitination, suggesting that endosomal processing of the viral capsid proteins is a prelude to ubiquitination. Furthermore, ubiquitination may be a signal for processing of the capsid at the time of virion disassembly. These studies suggest that the previously reported influences of the ubiquitin-proteasome system on rAAV-2 transduction are also active for rAAV-5 and provide a clearer mechanistic framework for understanding the functional significance of ubiquitination.